About Us
MedTech Innovator is the world’s largest accelerator for medical device, digital health, and diagnostic companies.

Fredrik Nyberg
Managing Director
LinkedInFredrik is the Managing Director Asia Pacific at MedTech Innovator, based in Singapore. He was most recently the Founding CEO of Asia Pacific Medical Technology Association (APACMed). Fredrik brings over 30 years of healthcare industry experience, 25 of which were spent in Asia-Pacific. He has held senior sales/ marketing, general management and strategic consulting positions. Fredrik serves as Board Member and advisor to several healthcare startup ventures and is a frequent speaker on the healthcare industry in Asia-Pacific. He holds a BSc from Gothenburg University, Sweden, and an MBA from Henley Management College, UK.

Glen Lim
Senior Program Director
LinkedInGlen is Senior Programs Director, Asia Pacific at MedTech Innovator, based in Singapore. He has led successful global programs at Enterprise Singapore’s Singapore Week of Innovation and Technology mega event (SWITCH), helping startups in access the Singapore Market and expand into Asia Pacific. Glen is also the founder of 2 startups and serves as a Venture Partner at Meet Ventures. In his free time, Glen mentors young first time startup founders in Polytechnics and Universities.

Nerissa Yu
Program Manager
LinkedInNerissa is the Program Manager for Asia Pacific at MedTech Innovator, based in Singapore. She fosters the development and growth of medtech startups. Her extensive program management background spans across public, private, and diverse industry sectors, bringing a dynamic range of expertise to her role. Previously, Nerissa led innovation-focused programs at a prominent government agency in Singapore, nurturing the startup ecosystem. Following that, at the Development Bank of Singapore (DBS), she designed and managed financial literacy programs for parents and children.

Jermaine Lok
Partnerships Manager
LinkedInJermaine is the Partnership Manager for Asia Pacific at MedTech Innovator, based in Singapore. She supports the strategic collaborations across the region. Before joining MedTech Innovator, Jermaine managed international partnerships and innovation programs at Enterprise Singapore, leading initiatives connecting startups, corporates, and government agencies across Asia and North America.

Bernice Tan
Marketing & Events Manager
LinkedInBernice is the Marketing and Events Manager for Asia Pacific at MedTech Innovator, based in Singapore. She is a seasoned Marketing & Events Professional renowned for her exceptional expertise in devising and implementing strategic initiatives that fuel growth. Bernice expertly manages various simultaneous events like networking, hybrid functions, and expos, showcasing her exceptional organizational skills and meticulous attention to detail for flawless coordination across all aspects of these diverse gatherings.

Don’t Miss Your Chance To Apply! Applications close 31 January 2025
Watch one of our info sessions from 2025, or view our FAQ’s for answers to common questions.
2025 Info Sessions
Info Session (APAC Focused) – 26 November 2024
Info Session (Australia Focused) – 3 December 2024
Info Session (China Focused) – 8 January 2024
Info Session (Japan Focused) – 9 January 2024
Info Session (South Korea Focused) – 14 January 2024


MedTech Innovator does not require equity and there is no application fee. However, companies are responsible for their travel costs related to attending pitch events and mandatory in-person events, where applicable.
MedTech Innovator seeks early to mid-stage start-ups in the medical device, diagnostic, or digital health/health IT spaces. We encourage those who are developing a product and/or service in the healthcare space along one of these verticals to apply.
Yes! MedTech Innovator encourages applications from companies at all stages of fundraising, from friends and family to seed and beyond. Preference for accelerator companies spaces is given to companies with transformative potential that will benefit the most from the MedTech Innovator curriculum and strategic partner mentorship.
MedTech Innovator evaluates all applications based on value, execution, and momentum. The company strength for each category is evaluated using multiple metrics. Please see below for the complete evaluation metrics table.

Here are the points we recommend including in your deck:
- Problem / unmet need: show image of impacted patients, caregivers, etc.
- Market information
- Standard of care
- Your solution / technology overview: include product photo or rendering and expected clinical outcomes
- Competitive advantage / differentiation
- Validation: pre-clinical, clinical, market
- Regulatory path (if applicable) and planned indication
- Value proposition / healthcare economics
- Business model, reimbursement (if applicable)
- IP status: include broadest claims sought
- Team: photos with logos below from companies they have worked for previously
- Advisors / partners: photos with logos below from companies they work for currently
- Key milestones / timeline for go-to-market: include funding required to achieve each milestone point
- Summary, including value proposition: show image of impacted patients or product
We have a limited number of slots available for companies to pitch online. Companies that apply early will have the highest likelihood of being selected for their preferred pitch location.
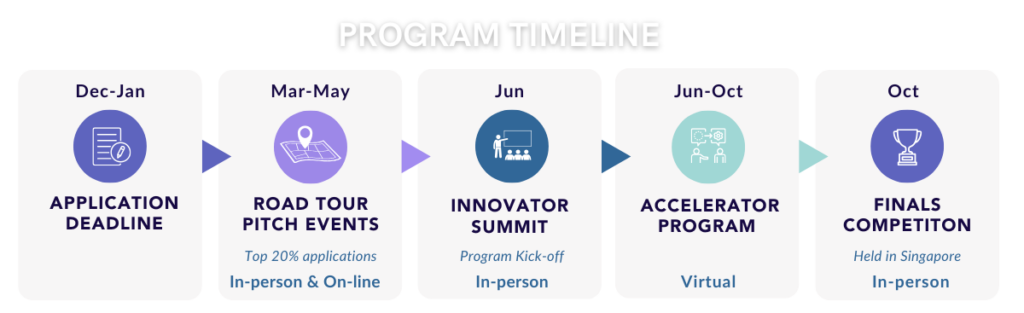
The MedTech Innovator Accelerator is virtual, except for two mandatory in-person events that start and end the program. The Innovator Summit will take place in Mountain View, California in June 2025 and the Showcase and Grand Finals will take place in Singapore in end October 2025.
Yes, you should definitely reapply! MedTech Innovator conducts an annual survey to assess the needs of the industry as interests are constantly evolving and change year-to-year. Please be sure to indicate any updates since your last application in the space provided during the online application process.
Value
• Product
• Customer Validation
• Business Model & Healthcare Economics
• Competition
• IP
• Market Opportunity
• Patient Benefit
Execution
• Team Strength & Execution
• Development Plan
• Responsive to Feedback
• Freedom to Operate
• Regulatory & Clinical Burden
• Existing Funding
• Resources to Execute
Momentum
• Stage of Development
• Evidence
• Time to Market
• Traction & Sales
• Partnerships & Relationships
• Time to Key Milestones
• Upcoming Funding
Here are the points we recommend including in your deck:
- Problem / unmet need: show image of impacted patients, caregivers, etc.
- Market information
- Standard of care
- Your solution / technology overview: include product photo or rendering and expected clinical outcomes
- Competitive advantage / differentiation
- Validation: pre-clinical, clinical, market
- Regulatory path (if applicable) and planned indication
- Value proposition / healthcare economics
- Business model, reimbursement (if applicable)
- IP status: include broadest claims sought
- Team: photos with logos below from companies they have worked for previously
- Advisors / partners: photos with logos below from companies they work for currently
- Key milestones / timeline for go-to-market: include funding required to achieve each milestone point
- Summary, including value proposition: show image of impacted patients or product
We have a limited number of slots available for companies to pitch online. Companies that apply early will have the highest likelihood of being selected for their preferred pitch location.
The MedTech Innovator Showcase is virtual, except for 1 mandatory in-person event that ends the program. The Showcase and Finals may take place at the MedTech Forum 2022 hosted by APACMed, subject to pandemic restrictions in 2022.
Yes, you should definitely reapply! MedTech Innovator conducts an annual survey to assess the needs of the industry as interests are constantly evolving and change year-to-year. Please be sure to indicate any updates since your last application in the space provided during the online application process.












